 She suggested that Americans should view these shots as an annual measure to protect oneself, in line with the annual flu shot.Īs with the flu, Pfizer/BioNTech, Moderna and Novavax have created versions of the Covid-19 vaccine to try to match the variant they believe will be circulating this autumn. More than 1.1 million people in the US have died from Covid-19, according to the Centres for Disease Control and Prevention (CDC).ĬDC director Mandy Cohen said last week in a podcast that she expects the shots – which still need to be authorised by the US Food and Drug Administration and recommended by the CDC – to be rolled out in the third or fourth week of September. David Kaslow said in June at the CDC meeting.The Covid-19 public health emergency ended in May, and the government has handed much of the duty of vaccinating America to the private sector. So in the fall, that would be the 2023-2024 formula, would be an XBB.1.5," the FDA's Dr. "The intent is to harmonize for all doses, all ages, same composition. Children down to 6 months still might be recommended to get two or three doses. Unlike vaccinations earlier in the pandemic, federal officials say they have been working in recent months to simplify eligibility for future rounds of shots, akin to the annual seasonal influenza shot.įor teens and adults, Americans would have their pick of any of the three updated vaccines.įor children as young as 2 years old, draft CDC vaccine recommendations presented in June would allow for a single new shot from either Pfizer or Moderna in order to be up to date. Who will be eligible to get the new COVID-19 vaccines? "While we cannot comment directly on timing, the FDA anticipates taking timely action to authorize or approve updated COVID-19 vaccines in order to make vaccines available this fall that meet our expectations for safety, effectiveness and quality," an FDA spokesperson said in a statement. Peter Marks, who had predicted the shots could be available in September. October would be later than previously forecasted by the FDA's top vaccines official Dr. That timeline could add up to new COVID-19 vaccines not being widely available until October, along the lines of what CDC Director Dr. This is needed to ensure liability protections for vaccinators as well as to guarantee insurance coverage and access to the new shots. "After their authorization or approval, ACIP will meet to make a recommendation outlining use of these updated vaccines this fall," CDC spokesperson Kathleen Conley told CBS News in a statement. However, the CDC does still plan to convene its Advisory Committee on Immunization Practices before issuing updated recommendations for the three new shots. The FDA is not expected to call another meeting of its Vaccines and Related Biological Products Advisory Committee before signing off on the new shots. 
"That's going to be concluded this month, with expectation for us to be delivering product by the end of September," Novavax's President of Research and Development Filip Dubovsky told investors on August 8. Novavax has yet to complete its submission for a new emergency use authorization for its updated vaccine, but plans to do so within the coming weeks. 
And we are ready with products already now," Pfizer's CEO Albert Bourla told investors on August 1. "What we expect is that we will have approval by the end of August. Pfizer and Moderna could be first to get the FDA's licensure, after finishing their submissions to the agency back in June. The FDA is expected to grant approval or emergency authorization to all three new COVID-19 vaccines over the next two months. commercial market: a green light from the FDA and new recommendations from the CDC. Two steps will still be needed before the new vaccines can make their debut in the U.S. While the new vaccines are expected to be ready by late September, it could be October before they're widely available for everyone who wants them. 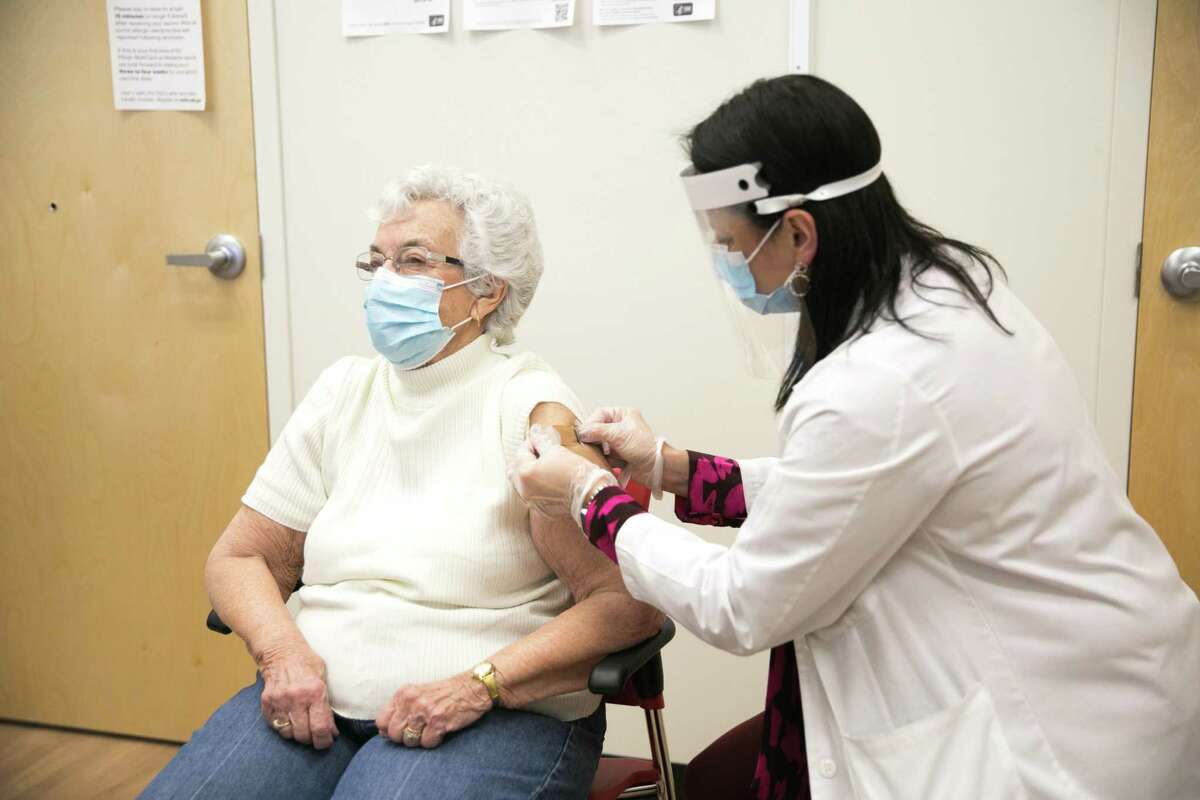
When will new COVID-19 vaccines be available? Eric Rubin, one of the FDA's vaccine advisers, said at the June meeting. 
XBB is the lineage right now, and there is good cross-protection, no matter what antigen is chosen, according to the data that we've been shown," Dr. "There doesn't seem to be any particular advantage to a bivalent vaccine.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
